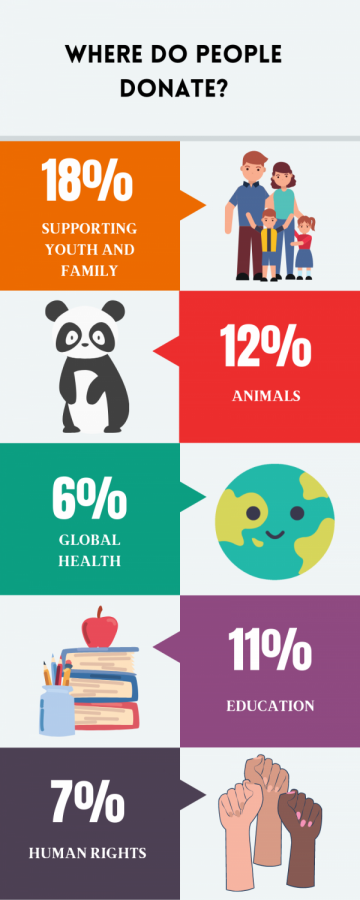
Light it up, up, up
A chemistry lesson in ionic salts
January 23, 2019
Each and every day in classrooms around Englewood High School, students are putting their lessons into practice. In Cassie Weasons chemistry class, students learned the production of an ionic salt using stoichiometry.
Each student carefully measured a compound of zinc and nitric acid and heated it with a bunsen burner. The groups will take measurements and show good lab techniques while gathering data sets.
Stoichiometry is a mouth full. It is a hard word to say and spell, but it is a big word that describes a simple process. Stoichiometry is the study of amounts of substances that are involved in reactions. You might be looking at the amounts of substances before the reaction. You might be looking at the amount of material that is produced by the reaction. Stoichiometry is all about the numbers, how they balance, how they react and how much stuff you have going into and out of the reaction.